VOLUNTARY FOOD RECALL NOTIFICATION
URGENT: FOOD RECALL NOTICE
PART 1: RECALL IDENTIFICATION
1.1 Recall Classification
Recall Type:
☐ Class I - Reasonable probability of serious adverse health consequences or death
☐ Class II - May cause temporary or medically reversible adverse health consequences
☐ Class III - Not likely to cause adverse health consequences
Recall Status:
☐ Firm-Initiated Voluntary Recall
☐ FDA-Requested Recall
Recall Number: [________________________________]
Recall Initiation Date: [__/__/____]
PART 2: RECALLING FIRM INFORMATION
2.1 Company Information
| Field | Information |
|---|---|
| Company Name | [________________________________] |
| Address | [________________________________] |
| City, State, ZIP | [________________________________] |
| Phone | [________________________________] |
| FEI Number | [________________________________] |
| FDA Registration Number | [________________________________] |
2.2 Recall Coordinator
| Field | Information |
|---|---|
| Name | [________________________________] |
| Title | [________________________________] |
| Phone | [________________________________] |
| [________________________________] | |
| Available Hours | [________________________________] |
PART 3: PRODUCT INFORMATION
3.1 Product Identification
| Field | Information |
|---|---|
| Product Name | [________________________________] |
| Brand Name | [________________________________] |
| Product Description | [________________________________] |
| Package Size(s) | [________________________________] |
| UPC Code(s) | [________________________________] |
| Product Code | [________________________________] |
3.2 Lot/Batch Information
| Lot Number | Production Date | Best By/Expiration Date | Quantity Produced |
|---|---|---|---|
| [________________________________] | [__/__/____] | [__/__/____] | [________________________________] |
| [________________________________] | [__/__/____] | [__/__/____] | [________________________________] |
| [________________________________] | [__/__/____] | [__/__/____] | [________________________________] |
| [________________________________] | [__/__/____] | [__/__/____] | [________________________________] |
3.3 Product Identification Marks
Location of Lot Code on Package: [________________________________]
Location of UPC Code: [________________________________]
Best By Date Format: [________________________________]
Other Identifying Marks: [________________________________]
PART 4: REASON FOR RECALL
4.1 Hazard Identification
Type of Hazard:
☐ Biological - Pathogen contamination
☐ Biological - Other: [________________________________]
☐ Chemical - Undeclared allergen
☐ Chemical - Foreign chemical contamination
☐ Chemical - Other: [________________________________]
☐ Physical - Foreign material
☐ Labeling - Incorrect/missing allergen declaration
☐ Labeling - Other: [________________________________]
☐ Other: [________________________________]
4.2 Specific Hazard Description
Contaminant/Hazard: [________________________________]
Potential Health Risk: [________________________________]
4.3 Allergen Information (if applicable)
Undeclared Allergen(s):
☐ Milk
☐ Eggs
☐ Fish: [________________________________]
☐ Shellfish: [________________________________]
☐ Tree Nuts: [________________________________]
☐ Peanuts
☐ Wheat
☐ Soybeans
☐ Sesame
☐ Other: [________________________________]
4.4 How Hazard Was Discovered
☐ Consumer complaint
☐ Internal testing
☐ Third-party laboratory testing
☐ Supplier notification
☐ FDA notification
☐ State agency notification
☐ Illness report
☐ Other: [________________________________]
Date Discovered: [__/__/____]
Discovery Details: [________________________________]
PART 5: DISTRIBUTION INFORMATION
5.1 Distribution Scope
Distribution Area:
☐ Nationwide
☐ Regional - States: [________________________________]
☐ Local - Area: [________________________________]
☐ International - Countries: [________________________________]
Distribution Dates: [__/__/____] to [__/__/____]
5.2 Distribution Channels
☐ Retail stores
☐ Wholesale distributors
☐ Food service/restaurants
☐ Online/direct to consumer
☐ Institutional (schools, hospitals)
☐ Other: [________________________________]
5.3 Quantity and Distribution Summary
| Category | Quantity |
|---|---|
| Total Quantity Produced | [________________________________] |
| Total Quantity Distributed | [________________________________] |
| Quantity in Company Possession | [________________________________] |
| Estimated Quantity Consumed | [________________________________] |
| Number of Direct Consignees | [________________________________] |
PART 6: CONSIGNEE NOTIFICATION
6.1 Recall Communication to Consignees
[LETTERHEAD]
Date: [__/__/____]
URGENT: FOOD RECALL NOTIFICATION
Dear Valued Customer:
[________________________________] is voluntarily recalling the following product(s) due to [________________________________].
PRODUCT(S) BEING RECALLED:
| Product Name | Lot Number(s) | UPC | Best By Date |
|---|---|---|---|
| [________________________________] | [________________________________] | [________________________________] | [________________________________] |
REASON FOR RECALL:
[________________________________]
HEALTH RISK:
[________________________________]
ACTION REQUIRED:
Please take the following immediate actions:
- ☐ Stop sale/distribution of the recalled product immediately
- ☐ Remove recalled product from shelves/inventory
- ☐ Segregate and quarantine all recalled product
- ☐ Do not destroy product until instructed
- ☐ Complete and return the enclosed response form by [__/__/____]
- ☐ Extend this recall notice to your customers/sub-consignees
PRODUCT DISPOSITION:
☐ Hold for pickup/return to [________________________________]
☐ Return to: [________________________________]
☐ Destroy according to the following instructions: [________________________________]
☐ Credit will be issued upon: [________________________________]
CONTACT INFORMATION:
For questions, contact:
Name: [________________________________]
Phone: [________________________________]
Email: [________________________________]
Please confirm receipt of this notice and your inventory status using the attached response form.
Sincerely,
[________________________________]
[________________________________]
[________________________________]
PART 7: CONSIGNEE RESPONSE FORM
Recall Response Form
Please complete and return by: [__/__/____]
Return to: [________________________________]
Fax: [________________________________] Email: [________________________________]
Consignee Information:
| Field | Information |
|---|---|
| Company Name | [________________________________] |
| Contact Person | [________________________________] |
| Address | [________________________________] |
| Phone | [________________________________] |
| [________________________________] |
Product Inventory Status:
| Product/Lot | Quantity On Hand | Quantity Distributed | Quantity Sold to Consumers |
|---|---|---|---|
| [________________________________] | [________________________________] | [________________________________] | [________________________________] |
☐ We have NONE of the recalled product in our possession
☐ We NEVER received any of the recalled product
☐ We have the recalled product and have taken the following action:
[________________________________]
Sub-Consignee Notification:
☐ N/A - We did not distribute to others
☐ We have notified all of our customers/sub-consignees
Number of sub-consignees notified: [____]
Authorized Signature: ________________________________________
Date: [__/__/____]
PART 8: PUBLIC NOTIFICATION (Press Release)
8.1 Press Release Template
FOR IMMEDIATE RELEASE
Date: [__/__/____]
Contact: [________________________________]
Phone: [________________________________]
Email: [________________________________]
[COMPANY NAME] VOLUNTARILY RECALLS [PRODUCT NAME] DUE TO [REASON]
[City, State] - [________________________________] is voluntarily recalling [________________________________] because [________________________________].
Product Details:
- Product Name: [________________________________]
- Package Size: [________________________________]
- UPC Code: [________________________________]
- Lot Numbers: [________________________________]
- Best By Dates: [________________________________]
- Distribution: [________________________________]
Reason for Recall:
[________________________________]
What Consumers Should Do:
Consumers who have purchased [________________________________] should [________________________________].
Consumers with questions may contact [________________________________] at [________________________________] during [________________________________].
Illness Reports:
[________________________________]
About [Company Name]:
[________________________________]
PART 9: FDA NOTIFICATION
9.1 Report to FDA
Reportable Recall Submission
☐ Submitted to FDA Recall Coordinator
☐ Submitted via FDA Safety Reporting Portal
☐ Submitted to local FDA District Office
FDA District Office: [________________________________]
Date Submitted: [__/__/____]
FDA Contact: [________________________________]
9.2 Information Provided to FDA
☐ Identity of the product
☐ Reason for the recall and hazard involved
☐ Extent of distribution (dates, geographic area, consignees)
☐ Copy of recall communication
☐ Number of direct accounts
☐ Proposed strategy for recall
PART 10: RECALL EFFECTIVENESS CHECKS
10.1 Effectiveness Check Plan
Level of Effectiveness Checks:
☐ Level A - 100% of consignees
☐ Level B - >10% of consignees
☐ Level C - 10% of consignees
☐ Level D - 2% of consignees
☐ Level E - No effectiveness checks
Method of Effectiveness Checks:
☐ Phone calls
☐ Personal visits
☐ Letters requiring response
☐ Combination
Frequency: [________________________________]
10.2 Effectiveness Check Log
| Date | Consignee | Contact Method | Response | Product Status | Notes |
|---|---|---|---|---|---|
| [__/__/____] | [________________________________] | [________________________________] | ☐ Confirmed ☐ No Response | [________________________________] | [________________________________] |
| [__/__/____] | [________________________________] | [________________________________] | ☐ Confirmed ☐ No Response | [________________________________] | [________________________________] |
| [__/__/____] | [________________________________] | [________________________________] | ☐ Confirmed ☐ No Response | [________________________________] | [________________________________] |
10.3 Recall Status Summary
| Date | Consignees Notified | Responses Received | Product Recovered | % Recovery |
|---|---|---|---|---|
| [__/__/____] | [____] | [____] | [________________________________] | [____]% |
PART 11: PRODUCT DISPOSITION
11.1 Disposition Method
☐ Return to manufacturer
☐ Destruction at consignee location
☐ Destruction at designated facility
☐ Reconditioning/reprocessing
☐ Conversion to non-food use
11.2 Destruction Documentation
| Date | Location | Quantity Destroyed | Method | Witness | Documentation |
|---|---|---|---|---|---|
| [__/__/____] | [________________________________] | [________________________________] | [________________________________] | [________________________________] | ☐ CoD Attached |
PART 12: RECALL TERMINATION
12.1 Recall Completion Criteria
☐ All known consignees have been notified
☐ Effectiveness checks completed to required level
☐ All recoverable product has been accounted for
☐ Product properly disposed of
☐ FDA notified of recall completion
12.2 Recall Termination Request
Date Recall Completed: [__/__/____]
Final Statistics:
| Metric | Number |
|---|---|
| Total Consignees Notified | [____] |
| Total Responses Received | [____] |
| Response Rate | [____]% |
| Total Product Recalled | [________________________________] |
| Total Product Destroyed | [________________________________] |
| Recovery Rate | [____]% |
IMPORTANT REGULATORY NOTES
Recall Classifications (21 CFR 7.41)
- Class I: Health hazard situations where there is a reasonable probability that use of the product will cause serious adverse health consequences or death
- Class II: Health hazard situations where use may cause temporary or medically reversible adverse health consequences
- Class III: Not likely to cause adverse health consequences
Required Actions Under 21 CFR Part 7
- Notify FDA of firm-initiated recalls
- Prepare and distribute recall communications
- Conduct effectiveness checks
- Maintain records of recall activities
FSMA Requirements (21 CFR 117.139)
- Written recall plan required for facilities with hazards requiring preventive controls
- Plan must include procedures for consignee notification, public notification (if appropriate), effectiveness checks, and product disposition
SOURCES AND REFERENCES
- 21 CFR Part 7 Subpart C - Recalls
- 21 CFR 117.139 - Recall Plan
- FDA Guidance: Initiation of Voluntary Recalls Under 21 CFR Part 7
- FDA Guidance: Public Warning and Notification of Recalls
- FDA Regulatory Procedures Manual Chapter 7
This template is provided for informational purposes only. Each recall must be tailored to the specific product and circumstances involved. Follow your facility's written recall plan and consult with FDA regulatory counsel.
Do more with Ezel
This free template is just the beginning. See how Ezel helps legal teams draft, research, and collaborate faster.
AI that drafts while you watch
Tell the AI what you need and watch your document transform in real-time. No more copy-pasting between tools or manually formatting changes.
- Natural language commands: "Add a force majeure clause"
- Context-aware suggestions based on document type
- Real-time streaming shows edits as they happen
- Milestone tracking and version comparison
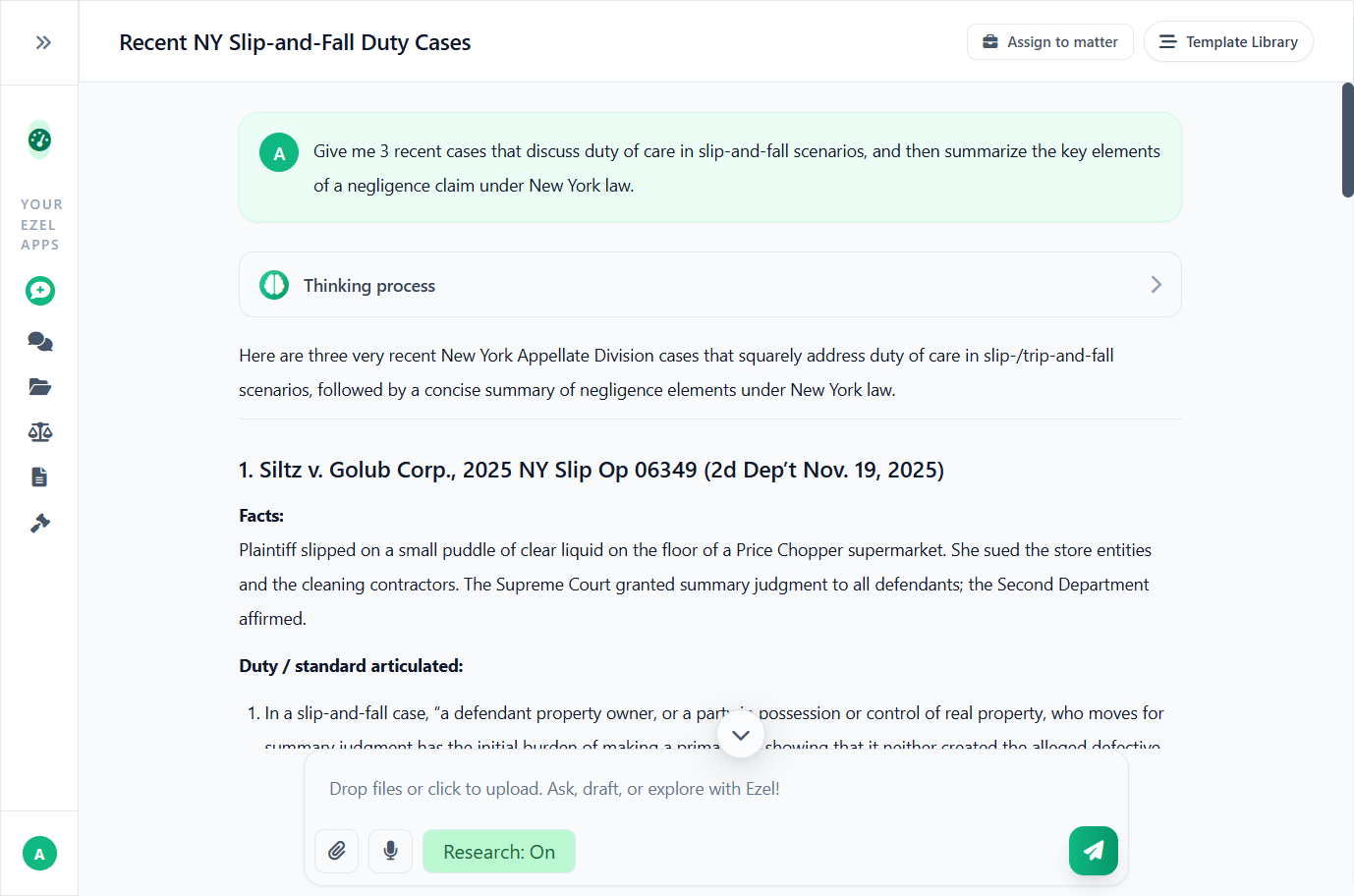
Research and draft in one conversation
Ask questions, attach documents, and get answers grounded in case law. Link chats to matters so the AI remembers your context.
- Pull statutes, case law, and secondary sources
- Attach and analyze contracts mid-conversation
- Link chats to matters for automatic context
- Your data never trains AI models
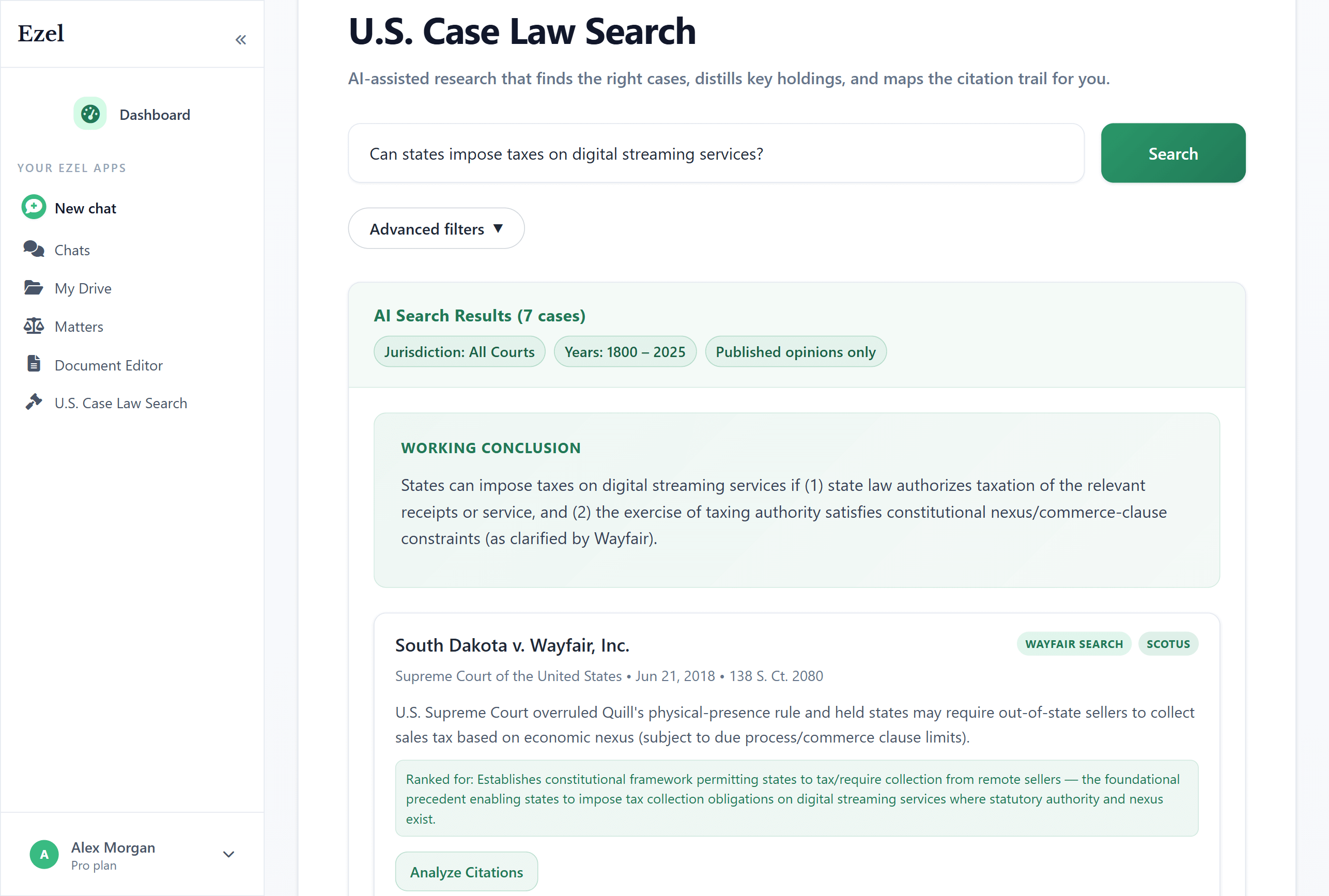
Search like you think
Describe your legal question in plain English. Filter by jurisdiction, date, and court level. Read full opinions without leaving Ezel.
- All 50 states plus federal courts
- Natural language queries - no boolean syntax
- Citation analysis and network exploration
- Copy quotes with automatic citation generation
Ready to transform your legal workflow?
Join legal teams using Ezel to draft documents, research case law, and organize matters — all in one workspace.