INFORMED CONSENT TO PARTICIPATE IN A RESEARCH STUDY
[INSTITUTION NAME]
Institutional Review Board Approval Required
STUDY INFORMATION
Study Title: [FULL STUDY TITLE]
Protocol Number: [PROTOCOL NUMBER]
Sponsor: [SPONSOR NAME]
Principal Investigator: [PI NAME], [CREDENTIALS]
Study Site: [SITE NAME AND ADDRESS]
24-Hour Contact: [PHONE NUMBER]
IRB Approval Date: [DATE]
IRB Approval Number: [IRB NUMBER]
[// GUIDANCE: Per 21 CFR 50.25 and 45 CFR 46.116, the consent form must include specific required elements. This template covers all basic elements and many additional elements. Customize for your specific study. The consent document may not include exculpatory language waiving the subject's legal rights or releasing the investigator/sponsor from liability for negligence.]
WHY ARE YOU BEING ASKED TO PARTICIPATE?
You are being asked to take part in a research study. Before you decide whether to participate, it is important that you understand why the research is being done and what it will involve. Please read the following information carefully and ask questions about anything you do not understand.
Your participation in this study is voluntary. You may choose not to participate, or you may withdraw at any time without penalty or loss of benefits to which you are otherwise entitled.
SECTION 1: PURPOSE OF THE STUDY
[// GUIDANCE: Per 21 CFR 50.25(a)(1) and 45 CFR 46.116(b)(1), describe that the study involves research, explain the purposes, expected duration, and procedures.]
This study is being conducted to:
[DESCRIBE THE PURPOSE OF THE RESEARCH IN PLAIN LANGUAGE]
Why is this research being done?
[EXPLAIN THE SCIENTIFIC RATIONALE]
How many people will participate?
Approximately [NUMBER] participants will take part in this study at [NUMBER] sites [nationwide/worldwide].
How long will I be in the study?
Your participation will last approximately [DURATION]. This includes [NUMBER] study visits over [TIMEFRAME].
SECTION 2: STUDY PROCEDURES
[// GUIDANCE: Per 21 CFR 50.25(a)(1), describe the procedures to be followed.]
If you agree to participate, you will be asked to:
Screening Visit
☐ [PROCEDURE 1]
☐ [PROCEDURE 2]
☐ [PROCEDURE 3]
Treatment/Study Period
☐ [PROCEDURE 1]
☐ [PROCEDURE 2]
☐ [PROCEDURE 3]
Follow-up Visits
☐ [PROCEDURE 1]
☐ [PROCEDURE 2]
Study Schedule Summary
| Visit | Timing | Duration | Procedures |
|---|---|---|---|
| Screening | [TIMING] | [DURATION] | [PROCEDURES] |
| Visit 1 | [TIMING] | [DURATION] | [PROCEDURES] |
| Visit 2 | [TIMING] | [DURATION] | [PROCEDURES] |
| Final Visit | [TIMING] | [DURATION] | [PROCEDURES] |
Randomization (if applicable)
☐ This study involves randomization
☐ You will be randomly assigned to receive either [STUDY DRUG/TREATMENT] or [PLACEBO/COMPARATOR]
☐ You will have a [X]% chance of receiving [TREATMENT A] and [X]% chance of receiving [TREATMENT B]
☐ Neither you nor the study doctor will know which treatment you receive (double-blind)
Experimental Procedures
The following procedures are experimental (not standard of care):
☐ [EXPERIMENTAL PROCEDURE 1]
☐ [EXPERIMENTAL PROCEDURE 2]
SECTION 3: RISKS AND DISCOMFORTS
[// GUIDANCE: Per 21 CFR 50.25(a)(2) and 45 CFR 46.116(b)(2), describe reasonably foreseeable risks or discomforts.]
Participation in this study may involve the following risks:
Risks of [STUDY DRUG/TREATMENT]
Common Side Effects (occurring in more than 10% of participants):
☐ [SIDE EFFECT 1]
☐ [SIDE EFFECT 2]
Less Common Side Effects (occurring in 1-10% of participants):
☐ [SIDE EFFECT 1]
☐ [SIDE EFFECT 2]
Rare but Serious Side Effects (occurring in less than 1% of participants):
☐ [SIDE EFFECT 1]
☐ [SIDE EFFECT 2]
Unknown Risks:
☐ There may be risks that are currently unknown
☐ You will be informed of any new information that may affect your willingness to continue
Risks of Study Procedures
Blood Draws:
☐ Pain, bruising, or bleeding at the needle site
☐ Fainting or lightheadedness
☐ Infection (rare)
[OTHER PROCEDURES]:
☐ [RISKS]
Reproductive Risks (if applicable)
☐ This study drug may harm an unborn baby
☐ If you are pregnant or become pregnant, you should not participate
☐ You must use effective birth control during the study and for [DURATION] after
☐ If you become pregnant during the study, notify the study team immediately
SECTION 4: POTENTIAL BENEFITS
[// GUIDANCE: Per 21 CFR 50.25(a)(3) and 45 CFR 46.116(b)(3), describe any benefits to the subject or others.]
Potential benefits to you:
☐ You may experience [POTENTIAL BENEFIT]
☐ There may be no direct benefit to you from participating
☐ [OTHER POTENTIAL BENEFITS]
Potential benefits to others:
☐ Information learned from this study may help future patients
☐ [OTHER BENEFITS TO SOCIETY/SCIENCE]
SECTION 5: ALTERNATIVES TO PARTICIPATION
[// GUIDANCE: Per 21 CFR 50.25(a)(4) and 45 CFR 46.116(b)(4), disclose appropriate alternative procedures or treatments.]
Instead of participating in this study, you could:
☐ [ALTERNATIVE TREATMENT 1]
☐ [ALTERNATIVE TREATMENT 2]
☐ Receive standard care without participating in research
☐ Choose not to receive any treatment
The study doctor will discuss these alternatives with you.
SECTION 6: CONFIDENTIALITY
[// GUIDANCE: Per 21 CFR 50.25(a)(5) and 45 CFR 46.116(b)(5), describe the extent to which confidentiality will be maintained.]
Your privacy is important to us.
☐ Your identity and personal health information will be kept confidential to the extent permitted by law
☐ Your research records may be reviewed by:
- The study sponsor ([SPONSOR NAME]) and its representatives
- The Food and Drug Administration (FDA)
- The Institutional Review Board (IRB)
- [OTHER REGULATORY AUTHORITIES]
☐ Your name will not appear in any publications resulting from this research
☐ You will be identified by a study code number, not your name
☐ Research records will be stored securely with limited access
☐ A description of this clinical trial will be available on www.ClinicalTrials.gov as required by law
HIPAA Authorization:
☐ Your protected health information may be used and disclosed for research purposes as described in the attached HIPAA Authorization form
SECTION 7: COSTS AND COMPENSATION
Costs to You
☐ There will be no cost to you for study drug/treatment
☐ There will be no cost to you for study-related procedures
☐ You may be responsible for costs of:
- [SPECIFY COSTS]
☐ Your insurance may be billed for standard care procedures
Compensation
☐ You will receive $[AMOUNT] for each completed study visit
☐ You will receive $[AMOUNT] for completing the entire study
☐ Compensation schedule:
| Visit | Compensation |
|-------|--------------|
| [VISIT] | $[AMOUNT] |
☐ Compensation will be prorated if you withdraw early
☐ You will receive a 1099 tax form if compensation exceeds $600
☐ You will not receive compensation for participating
SECTION 8: RESEARCH-RELATED INJURY
[// GUIDANCE: Per 21 CFR 50.25(a)(6), describe compensation and medical treatment available if injury occurs for research involving more than minimal risk.]
If you are injured as a result of participating in this study:
☐ You will receive medical treatment for the injury
☐ [INSTITUTION/SPONSOR] will pay for the cost of treating research-related injuries
☐ [INSTITUTION/SPONSOR] does not have a program to provide additional compensation for research-related injuries
☐ [DESCRIBE ANY COMPENSATION POLICY]
☐ You do not waive any legal rights by signing this consent form
Report any research-related injury to:
[CONTACT INFORMATION]
SECTION 9: VOLUNTARY PARTICIPATION AND WITHDRAWAL
[// GUIDANCE: Per 21 CFR 50.25(a)(8), state that participation is voluntary and describe consequences of withdrawal.]
Your participation is completely voluntary.
☐ You may choose not to participate without penalty
☐ You may withdraw at any time without penalty or loss of benefits
☐ Withdrawing will not affect your medical care outside the study
☐ You will not lose any benefits to which you are otherwise entitled
The investigator may stop your participation if:
☐ It is in your best medical interest
☐ You do not follow study instructions
☐ The study is stopped
☐ [OTHER REASONS]
If you withdraw:
☐ Data already collected may continue to be used
☐ You may be asked to complete a final safety visit
☐ [OTHER CONSEQUENCES]
SECTION 10: NEW INFORMATION
[// GUIDANCE: Per 21 CFR 50.25(b)(5), describe how new findings will be communicated.]
☐ You will be informed of any new information that may affect your willingness to continue participating
☐ You may be asked to sign a new consent form if significant new information becomes available
SECTION 11: NUMBER OF PARTICIPANTS
Approximately [NUMBER] people will take part in this study.
SECTION 12: CLINICALTRIALS.GOV STATEMENT
[// GUIDANCE: Per 42 U.S.C. § 282(j)(3)(A)(iii) / 21 CFR 50.25(c), include this statement for applicable clinical trials.]
A description of this clinical trial will be available on http://www.ClinicalTrials.gov, as required by U.S. Law. This website will not include information that can identify you. At most, the website will include a summary of the results. You can search this website at any time.
SECTION 13: CONTACT INFORMATION
For questions about the study:
Principal Investigator: [NAME]
Phone: [PHONE]
Email: [EMAIL]
For questions about your rights as a research participant:
Institutional Review Board: [IRB NAME]
Phone: [PHONE]
Email: [EMAIL]
For medical emergencies:
24-Hour Number: [PHONE]
SECTION 14: CONSENT
Statement of Person Obtaining Consent
I have explained the research to the participant and answered all questions. I believe the participant understands the information in this consent form and freely gives consent to participate.
Signature of Person Obtaining Consent: ______________________________________
Printed Name: [NAME]
Date: ______________
Statement of Participant
☐ I have read (or have had read to me) this entire consent form
☐ I have had the opportunity to ask questions, and all my questions have been answered to my satisfaction
☐ I understand the information provided about the study, including the purpose, procedures, risks, benefits, alternatives, and my rights
☐ I voluntarily agree to participate in this research study
☐ I understand that I will receive a signed copy of this consent form
☐ I understand that my participation is voluntary and I may withdraw at any time
Participant Signature: ______________________________________
Printed Name: [NAME]
Date: ______________
Time: ______________
Legally Authorized Representative (if applicable)
If the participant is unable to provide consent, a legally authorized representative may consent on their behalf.
Representative Signature: ______________________________________
Printed Name: [NAME]
Relationship to Participant: [RELATIONSHIP]
Date: ______________
Reason participant cannot consent: [REASON]
Witness (if required)
Witness Signature: ______________________________________
Printed Name: [NAME]
Date: ______________
Impartial Witness (for participants who cannot read)
I witnessed the accurate reading of the consent form to the participant, who had the opportunity to ask questions. I confirm that the participant freely gave consent.
Witness Signature: ______________________________________
Printed Name: [NAME]
Date: ______________
OPTIONAL CONSENT SECTIONS
Future Use of Samples/Data
☐ I agree that my samples may be stored for future research
☐ I do NOT agree to future use of my samples
Signature: ______________________________________ Date: ______________
Genetic Research (if applicable)
☐ I agree to participate in the genetic research portion of this study
☐ I do NOT agree to participate in the genetic research portion
Signature: ______________________________________ Date: ______________
Contact for Future Studies
☐ I agree to be contacted about future research studies
☐ I do NOT agree to be contacted about future studies
Signature: ______________________________________ Date: ______________
[// GUIDANCE: This consent form must be approved by an IRB before use. The consent process should allow adequate time for the subject to consider participation. A copy of the signed consent must be given to the participant. Retain consent forms per regulatory requirements (typically at least 2 years after study completion or longer if required by regulations).]
Do more with Ezel
This free template is just the beginning. See how Ezel helps legal teams draft, research, and collaborate faster.
AI that drafts while you watch
Tell the AI what you need and watch your document transform in real-time. No more copy-pasting between tools or manually formatting changes.
- Natural language commands: "Add a force majeure clause"
- Context-aware suggestions based on document type
- Real-time streaming shows edits as they happen
- Milestone tracking and version comparison
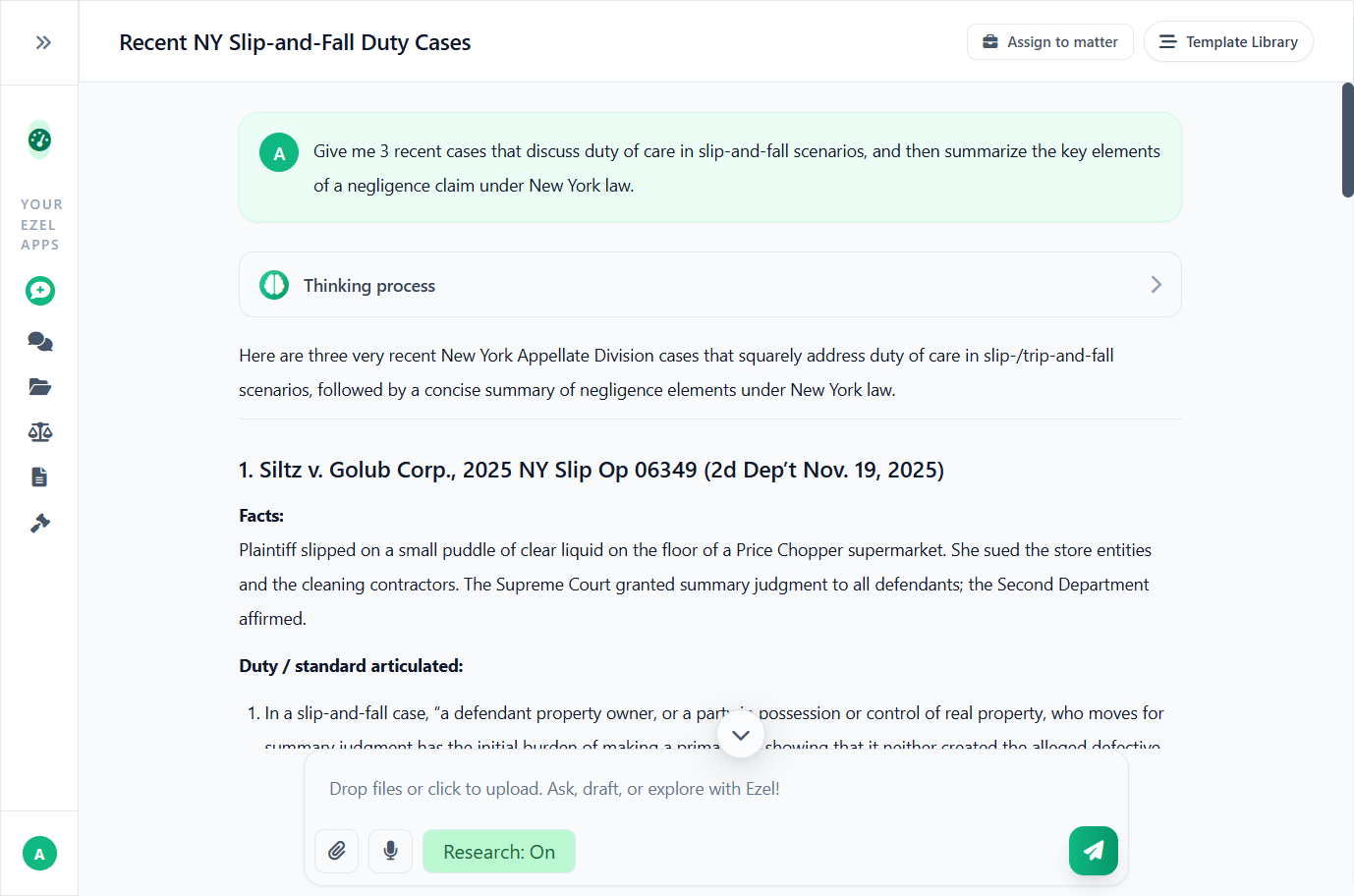
Research and draft in one conversation
Ask questions, attach documents, and get answers grounded in case law. Link chats to matters so the AI remembers your context.
- Pull statutes, case law, and secondary sources
- Attach and analyze contracts mid-conversation
- Link chats to matters for automatic context
- Your data never trains AI models
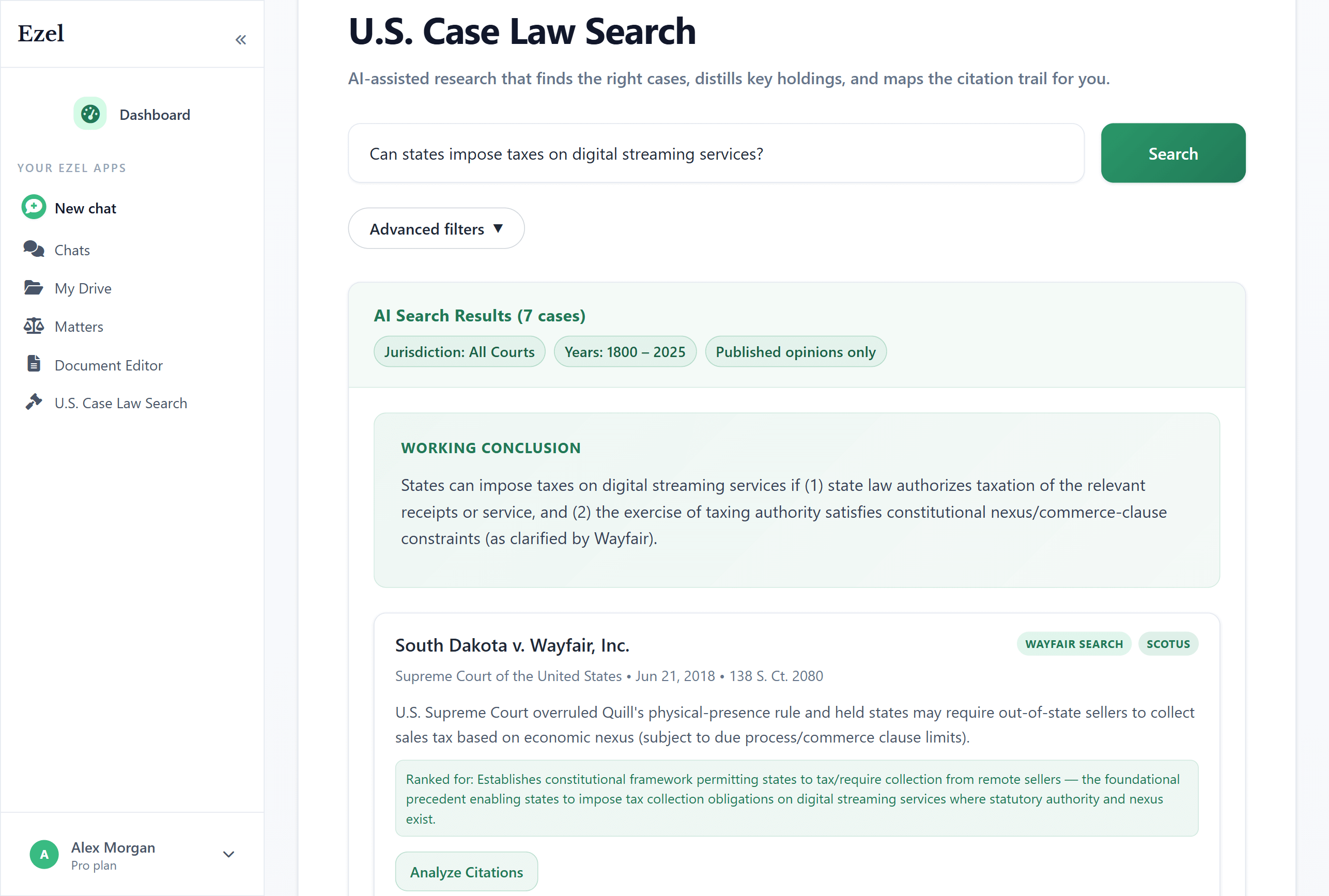
Search like you think
Describe your legal question in plain English. Filter by jurisdiction, date, and court level. Read full opinions without leaving Ezel.
- All 50 states plus federal courts
- Natural language queries - no boolean syntax
- Citation analysis and network exploration
- Copy quotes with automatic citation generation
Ready to transform your legal workflow?
Join legal teams using Ezel to draft documents, research case law, and organize matters — all in one workspace.